How can we use methane to make industries more sustainable?
Carbon dioxide and methane are highly potent greenhouse gases and are two of the leading drivers of climate change. However, through a process called dry reforming, we can combine carbon dioxide and methane to create ‘syngas’, which can be used to make fuels and plastics. Unfortunately, current dry reforming methods are inefficient and rely on fossil fuels. To address this, Professor Akshat Tanksale and his team at the RECARB Hub at Monash University, Australia, are designing a new reactor to improve efficiency and sustainability.
Talk like a chemical engineer
Biogas — gaseous fuel, such as methane, produced by the fermentation of organic matter
Catalyst — a substance that increases the rate of a chemical reaction without undergoing permanent change itself
Dry reforming — a catalytic process that converts methane and carbon dioxide into syngas
Global warming potential — a measure of how much heat a greenhouse gas traps in the atmosphere
Magnetic induction heating — the use of an alternating magnetic field to generate heat within electrically conductive metals
Syngas — short for synthesis gas: a mixture of carbon monoxide and hydrogen, produced industrially
In order to mitigate the effects of climate change, we must rapidly reduce the levels of greenhouses gases in our atmosphere. Although carbon dioxide is the main driver of climate change, other greenhouse gases also contribute to the problem. For example, methane is responsible for around 30% of the rise in global temperatures since the Industrial Revolution of the 1800s.
Although methane is produced naturally by volcanoes and the decomposition of organic matter in wetlands, more than 60% of methane emissions into the atmosphere are caused by human activities such as cattle farming, waste management and fossil fuel extraction.
In contrast to carbon dioxide, which stays in our atmosphere for hundreds of years, methane tends to stick around for a much shorter period of time – about 12 years. Even so, methane can trap 80 times more heat than carbon dioxide over a 20-year period. Due to its short atmospheric lifetime and high global warming potential, reducing methane emissions could be the quickest way to slow global warming.
This is what motivates Professor Akshat Tanksale and his team at the ARC Research Hub for Carbon Utilisation and Recycling (RECARB Hub) at Monash University. They are designing a new reactor that uses a process known as dry reforming to transform methane and carbon dioxide into useful products – mitigating climate change and providing valuable resources at the same time.
“In urban areas, methane produced from the decomposition of organic waste at landfill sites can be avoided by separating organic waste from solid waste at the source,” says Akshat. “The organic waste can then be sent to anaerobic digesters, oxygen-free tanks that use microorganisms to break down organic waste and produce biogas (a mixture of methane and carbon dioxide).” Likewise, in rural and farming areas, manure can be sent to anaerobic digesters to produce biogas. “The biogas can then be upgraded to syngas using the technology we have developed,” explains Akshat.
Dry reforming
“When methane and carbon dioxide are heated together in the process of dry reforming, they react to form syngas, a mixture of hydrogen and carbon dioxide,” continues Akshat. Syngas is an incredibly valuable product that can be used to make fuels, plastics and a wide range of chemicals, and is already used in many industries. “So, instead of letting methane and carbon dioxide escape into the atmosphere and worsen climate change, we can reuse them as raw materials,” explains Akshat. “It’s recycling greenhouse gases into useful products, rather than throwing them away.”
Unfortunately, current dry reforming processes are inefficient and can do more harm than good in terms of climate change. “Dry reforming is typically carried out in large industrial reactors that are heated by burning fossil fuels,” says Akshat. “This creates a big problem: even though you’re transforming greenhouse gases, you’re also producing more emissions just to keep the reactor hot.”
Current methods have other issues too. “The catalysts inside the reactors get damaged over time through the build-up of carbon, which stops them working properly,” explains Akshat. “Additionally, the heat inside these reactors is uneven, which wastes energy and shortens the system’s lifespan.” In theory, dry reforming is a great idea, but in practice, the process is inefficient and unsustainable.
A new reactor
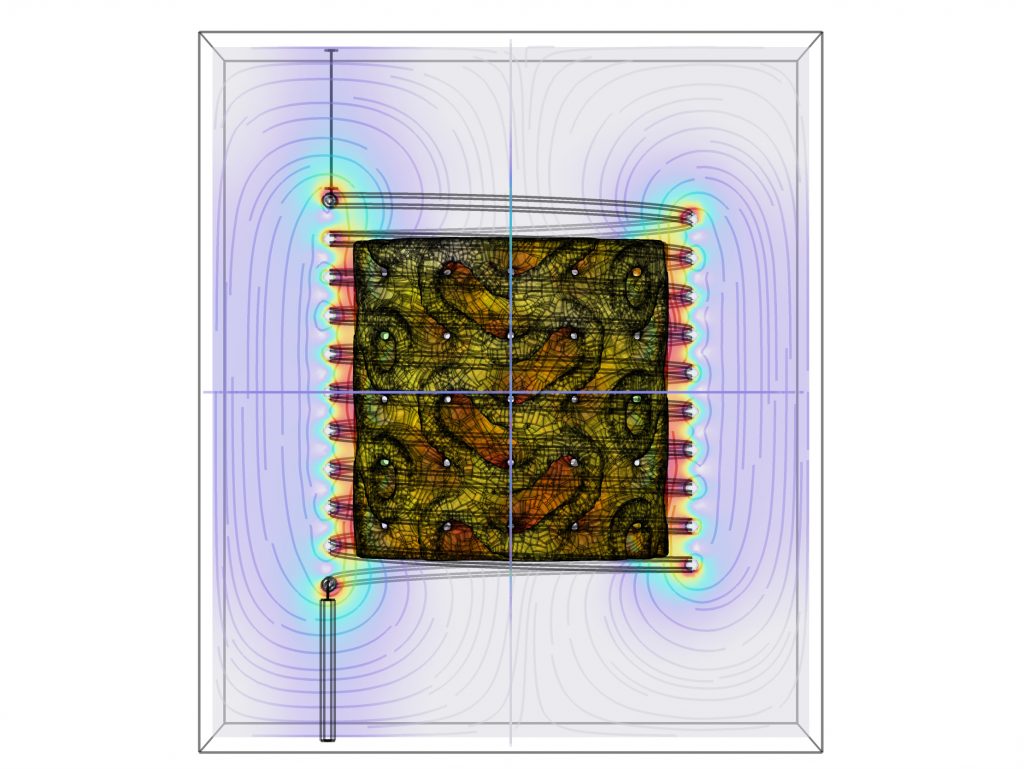
To turn dry reforming into a viable strategy for mitigating climate change, Akshat and his team have designed a new reactor that addresses the current inefficiencies. “We heat our reactor using electricity through a process called magnetic induction heating – similar to how induction stoves work,” says Akshat. “The electricity we use can be generated from renewable sources, such as solar and wind, which means that we can run this very hot chemical reaction without producing extra carbon emissions.”
“We also use carefully-designed, 3D-printed metal structures that sit inside the reactor,” says Akshat. “These structures allow for more even heating and gas flow, making the reaction faster, more efficient and more stable.” This also avoids carbon build-up on the catalysts, so the reactor can work for longer and with less wasted energy.
Into industry
The team’s next goal is to test the reactor under the conditions found in existing syngas industries. “This means it must be able to run for longer periods of time, produce higher quantities of syngas, and process gas mixtures that more closely resemble real-world sources such as biogas, which is produced by the fermentation of organic matter in waste systems, agriculture and landfills,” says Akshat. “We want to continue improving our reactor design so that it uses even less energy and works reliably for thousands of hours, not just in the lab, but as an important part of many different industries.”
Reference
https://doi.org/10.33424/FUTURUM684
Many industries, such as chemical, plastics and fuel manufacturing, already rely heavily on syngas, so the team’s new reactor will slot right in to their existing processes, meaning they can produce the same products with much lower emissions. “Our reactor is compact and modular, so companies could install it more easily than traditional systems,” explains Akshat. “It could even be placed near methane sources such as landfills and biogas plants, reducing transport and energy costs, allowing these industries to become more sustainable and save money at the same time.”
“Climate change is especially hard to tackle in these industries that rely on extremely high temperatures, but our work shows that even these processes can be powered by clean electricity,” continues Akshat. “This is not a golden bullet for climate change, but it would be a big step toward a cleaner industry and a more sustainable future.”
 Professor Akshat Tanksale
Professor Akshat Tanksale
Department of Chemical and Biological Engineering, Monash University;
Deputy Director, ARC Research Hub for Carbon Utilisation and Recycling
Fields of research: Chemical engineering; catalysis; carbon capture and utilisation
Research project: Creating an electrified reactor to make syngas production more efficient and environmentally friendly
Funders: Australian Research Council (ARC); Industry partners
About methane reduction
The field of methane reduction involves researching how to prevent or mitigate methane emissions into the atmosphere to minimise their contribution to global warming. “Research in methane reduction combines chemistry, engineering, materials science, energy systems and climate science,” says Akshat. “It’s a growing field with huge opportunities, giving young scientists and engineers a chance to be part of the global transition to a low-carbon future.”
Methane is a potent greenhouse gas, but it does not stay in the atmosphere as long as other gases, such as carbon dioxide. “This means that reducing methane emissions can have a fast and meaningful impact on climate change,” says Akshat. “As a researcher, it’s incredibly motivating to consider that your work can make a real difference within just a few years.”
It is important to consider methane’s value as a resource, which can also help build the financial incentive to use it rather than lose it. “When we convert methane into fuels, chemicals or hydrogen, we’re solving an environmental challenge while supporting the industries people rely on,” says Akshat. “We have to think beyond the impact on the climate; if a system is too expensive, it won’t be used.”
However, even if the theory is sound, its application can be another matter. “Reactions are complex and involve high temperatures,” explains Akshat. “This means that they require a lot of energy and put a lot of stress on the materials involved.” There is, therefore, a strong focus on designing systems to be efficient, durable and safe – not to mention collaborating with engineers and industry partners to ensure solutions work at scale.
There are plenty of exciting emerging technologies for the next generation of researchers to tackle. “Future researchers will work on electrified reactors, new catalysts, renewable energy integration and circular carbon technologies,” says Akshat. “They won’t just be studying climate change; they’ll be actively building the solutions.”
Pathway from school to methane reduction
Akshat says that studying chemistry, physics and mathematics is vital for entering the field of methane reduction. He also recommends studying environmental science, engineering or computer science if the you have the opportunity to do so.
At university, relevant degree courses include chemical engineering, chemistry, materials science, environmental engineering, energy engineering and physics. Akshat also suggests learning coding, data science or modelling, which are becoming increasingly important in his field of research.
Explore careers in methane reduction
The scope of careers related to methane reduction is broad. “Graduates can work as research scientists or engineers, designing cleaner reactors and catalysts,” says Akshat. “There are also roles in clean energy companies, chemical and fuel industries, climate tech start-ups, as well as policy and environmental regulation.”
Akshat recommends building a habit of learning beyond the classroom. He recommends exploring the websites of NASA, the International Energy Agency and Our World in Data.
Akshat also recommends watching educational videos on YouTube. Some good channels to try include Kurzgesagt, Veritasium and Real Engineering.
Meet the team
 Dr Hamza Asmat
Dr Hamza Asmat
Research Fellow
Growing up, I was fascinated by how energy and chemical systems work. I loved experimenting and trying to understand the ‘why’ behind everyday processes. This curiosity led me to study chemical engineering, where I could combine my interest in science with practical problem solving.
Over time, I became more aware of the environmental challenges we face. I saw the potential for technology to create a meaningful change. Now, I aim to develop innovative solutions that improve energy efficiency, reduce emissions and contribute to a cleaner, more sustainable future.
This project formed the core of my PhD. I developed the fundamental catalyst and reactor concepts for methane reduction through electrified reforming. I now continue this work as a postdoctoral researcher, leading its scale-up and optimisation.
I love being involved in research that tackles the toughest challenges in energy transition. And I enjoy collaborating with a talented team, learning from diverse perspectives and collectively pushing the boundaries of what is possible.
Outside of work, I spend time with my family. We often go for walks or enjoy simple activities at home. I also find cooking and listening to music help me return to work feeling refreshed, motivated and balanced.
Hamza’s top tip
Be patient and persistent. Meaningful impact takes time but is incredibly rewarding.
 Ashwin Hatwar
Ashwin Hatwar
3rd Year PhD student
As a teenager, I was deeply interested in the problem of climate change – and the search for solutions. I regularly took part in science exhibitions and presented models that demonstrated the science behind climate change. The sense of urgency, and my curiosity about solutions, motivated me to pursue research focused on developing sustainable technologies.
My role in this project is to develop and study this new type of reactor. I focus on designing the reactor, selecting and testing catalysts, and understanding how heat and electricity can be used more effectively.
Curiosity and persistence have been essential in my research. Experiments often require patience and an ability to learn from failure. Adaptability and clear communication have also allowed me to work effectively with others and adjust when research takes unexpected directions.
I love feeling that my work connects directly to a global challenge. Every experiment feels meaningful. I also enjoy the excitement of discovery – seeing an idea work in the lab and knowing it could one day make a real difference is incredibly rewarding.
Outside of work, I like watching movies, going out with friends and cooking. I stay active through sports like badminton and cycling, which help me clear my mind and recharge.
Ashwin’s top tip
Even small contributions matter. We can all be part of a global effort to create a more sustainable future.
 Mohammad Arfin
Mohammad Arfin
3rd Year PhD student
I’ve always been fascinated by how physical and chemical processes work and shape the world around us. This curiosity led me towards chemical engineering, and eventually to research, where I could focus on the problems most in need of solutions.
Climate change is one of the defining challenges of our time. Developing more sustainable chemical processes to help address this challenge feels both meaningful and rewarding. This has shaped my current work.
In this project, I focus on developing scalable, electrified dry reforming processes. While research shows the potential of these technologies, scaling them remains a major challenge. I help bridge that gap by focusing on real-world implementation strategies.
Curiosity has been central to my research approach: always asking ‘why’ and ‘how’. Staying aware of the latest developments in the field is also important to help me identify ideas that are both innovative and practically achievable.
This research brings a strong sense of purpose. I love the idea of creating real ‘impact through research’, and it is motivating to see our work steadily moving in that direction.
I’m a family person. I like staying connected with my family, but also value some moments alone to reset and recharge.
Arfin’s top tip
View challenges as learning opportunities to help build resilience and long-term confidence.
Do you have a question for Akshat, Hamza, Ashwin or Arfin?
Write it in the comments box below and they will get back to you. (Remember, researchers are very busy people, so you may have to wait a few days.)










0 Comments