Concrete change: the innovative chemistry of sustainable cement
The global cement industry is gigantic, so finding ways to reduce its impact on the climate is essential for a sustainable future. At the University of Sheffield in the UK, Dr Theodore Hanein and his team are investigating alternative methods for cement production. This includes replacing raw materials with waste products from other industries and reducing the carbon dioxide released during cement manufacture.
TALK LIKE A MATERIALS SCIENTIST AND PROCESS ENGINEER
By-product — unnecessary product produced during a process
Carbon capture and storage (CCS) — the capture and storage of CO2 emissions to prevent the gas entering the atmosphere
Cement — a powdery substance made from lime and clay, that can be used to make concrete and mortar
Circular economy — an economic system based on the reuse and reutilisation of materials and products
Clinker — the hard product made from processing raw materials, such as limestone and clay, at high temperatures (around 1500 °C). Making clinker is the first step in the conventional cement-making process
Concrete — a building material made of cement, water and aggregates (for example, sand and gravel) that hardens into a stone-like mass
Decarbonise — reduce or eliminate CO2 emissions from a process
Lime — a powder created by heating limestone, and an intermediary substance in the creation of cement
Limestone — a naturally occurring rock, mostly composed of calcium carbonate, used as a core raw material for the manufacture of cement
Mortar — a material made of cement, sand and water used in construction to bind bricks or stones together
“Cement is the most manufactured commodity in the world,” says Dr Theodore (Theo) Hanein, from the University of Sheffield. “Current demand is at 4 billion tonnes every year, which is more than one kilogram of cement every day for every person on the planet!” Cement is vital for society as it is a core material of concrete and mortar, which are essential components in the buildings and infrastructure that surround us. We cannot live without it.
However, all this production has a serious impact on the planet. “Cement production requires vast amounts of natural resources, and is responsible for 8% of our carbon dioxide (CO2) emissions, which are changing the climate,” explains Theo. In line with global climate commitments, governments are investing in ways to reduce CO2 emissions, and the cement industry is a prime area of interest. “The UK cement industry is set to cut 4.2 megatons (Mt) of CO2 emissions per year by 2050 – and about half of these savings could be through resource efficiency,” he says.
The chemistry of cement
It is the use of resources, especially raw materials, that forms the current focus of Theo’s research. “Unlike other manufacturing processes that emit CO2 through their use of energy, emissions from cement production are mainly from the conversion of the raw material into clinker, a key process in cement manufacture,” he says. While some energy-intensive industries can decarbonise through switching to renewable energy inputs, it is not so simple for cement. By far, the most common type of cement produced today – and for the past 200 years – is Portland cement, which relies on the following reaction to begin production:
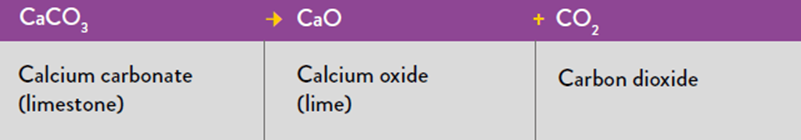
“Not only does this emit CO2, but almost half of the limestone’s solid mass is lost in the process,” says Theo. “The UK cement industry uses about 12.5 Mt of limestone every year, so we’re interested to see if this natural raw material can be substituted with by-products from other sectors.” This could be by-products from steel manufacturing or mineral mining, or even the reprocessing of old cement from construction or demolition sites. Developing this process would have the double benefit of cutting down cement the emissions from cement production, as well as using the waste produced by other sectors.
The contamination challenge
The main issue with such reprocessing efforts is that any alternative materials will contain other elements that could change the underlying chemistry of the cement manufacturing process and the end product. “The effects of these contaminants across the service life of the new cement needs to be studied and understood completely to ensure that the cement can be used safely,” explains Theo. Cement that has previously been used in construction, for instance, has generally been mixed with other materials, such as sand and gravel, to give it the properties needed, but that can interfere with the repurposing process which needs to be done for the cement to be reused.
A particular project of Theo’s, called FeRICH, is investigating this challenge when utilising by-products from steel manufacture. “While these by-products contain the key elements that are essential to cement production, they also have an unusually high amount of iron,” says Theo.
“FeRICH aims to replace natural raw materials used in traditional Portland cement by upcycling iron-rich by-products, but our understanding of the reactions that take place when we do this is still incomplete.” The introduction of iron leads to the production of calcium (alumino)ferrites (compounds made from iron and one or more metallic element), such as Ca2(Al,Fe)2O5. Theo is interested in learning what this means for the qualities of the end product, especially its durability and how it reacts with water.
“The FeRICH project will establish the best cement-making conditions to achieve maximum efficiency in both production and performance,” says Theo. “Calcium (alumino)ferrites containing contaminants (also known as dopants) could, potentially, also give the cement electromagnetic properties, and we’ll be examining how this could bring benefits or disadvantages to the end product.” By establishing these traits of ferrite-rich cement, the project can help reduce the environmental impact of both the cement and steel industries in the UK. This would also increase their economic competitiveness in the global market, bringing a boost to the UK and, as other nations follow suit, the rest of the world’s cement and steel industries.
CO2 and the circular economy
While these processes can help increase the efficiency of cement production, the production of CO2 as a by-product remains inevitable. To address this, a lot of investment is taking place into carbon capture and storage (CCS) technologies, and Theo has developed CCS technology to separate CO2 from limestone without it being released as a gas. “CCS may be able to capture CO2 from the exhausts of cement kilns, but we need to find a safe way to then store it,” he says. There are various projects around the world that are finding ways to pipe compressed CO2 underground or underwater – for example, into empty oil or gas fields (areas where these natural resources have been found and extracted for use as a fuel). However, Theo thinks that using chemical reactions to change the CO2 into something else is likely a better solution. “Currently, the safest and most developed method of CCS is probably via mineralisation,” he explains. This involves the reaction of CO2 with other materials to produce solid minerals. “It would be most beneficial if the generated minerals could be reused in the construction or mining industries, to avoid the storage of large amounts of material.”
Reference
https://doi.org/10.33424/FUTURUM402
A running theme across a lot of Theo’s research involves connecting different sectors and finding ways where the waste products of one can be a valuable resource for another. This feeds into a broader emerging concept known as the circular economy. Currently, much of our economy is based on linear processes: we extract a raw material, process it and use it, and then discard the waste products. This is a serious problem for several reasons. For one, the world’s resources are finite, and for another, waste takes up space and releases contaminants into our environment and atmosphere. Finding ways to break this trend, through repurposing one industry’s waste as the raw materials of another, simultaneously solves the issues of extraction of the materials and the disposal of waste. It is a key concept for a truly sustainable future. “The UK is working towards having a circular economy, but we’re certainly not there yet,” says Theo. “We need to redesign our processes for sustainability and to accelerate industry take-up.” Theo is taking a key role in this, using his innovative research to further understand and scale up the uptake of novel cements across the UK.
 Dr Theodore Hanein
Dr Theodore Hanein
Department of Materials Science and Engineering, University of Sheffield, UK
Fields of research: Cement Materials Science, Chemical Process Engineering, Thermodynamics
Research project: Investigating the chemical processes and qualities involved in alternative cement manufacture, and how this could bring benefits to the UK’s environmental targets and economic goals
Funder: UK Research and Innovation (UKRI EPSRC grant reference EP/W018810/1)
ABOUT MATERIALS SCIENCE AND PROCESS ENGINEERING
We have used the Earth’s resources to build the infrastructures that underpin our society, but the way we use these materials needs a major overhaul to ensure a sustainable future. Investigating the chemical and thermodynamic processes behind manufacturing – and, in particular, how to make these processes more efficient and circular – forms the core of Theo’s research. He explains more about his field and its importance.
“To make any device, structure or product, you need the right materials. Materials science involves learning about what things are made of, and what properties make them behave as they do. It also involves applying this knowledge to improve these processes and properties.
“Materials science and process engineering requires the integration of various fields. We incorporate knowledge from chemistry, physics, mathematics and biology with chemical engineering to help address society’s most pressing challenges.
“I find my research highly rewarding because it feels important. I am contributing to developing sustainable materials and processes, helping to solve the most pressing challenge of our time: climate change.
“Most of our processes were not designed with sustainability or the circular economy in mind. We need to redesign and develop new processes for an environmentally friendly world. Sticking-plaster methods such as CCS are useful for the short term, but, in the long term, we need to re-engineer our processes and society as a whole for true circularity.”
Pathway from school to materials science and process engineering
• At school and post-16, subjects such as physics, chemistry and mathematics are favourable to go on to a relevant degree.
• Theo recommends university courses or modules in materials science and engineering, chemical engineering, product design engineering, computer science, environmental engineering, chemistry and physics.
Explore careers in materials science and process engineering
• The University of Sheffield’s Department of Materials Science and Engineering hosts a variety of open days, summer schools and other STEM activities aimed at secondary school students.
• The Institute of Materials, Minerals and Mining (IOM3) has a variety of outreach materials for students of all ages and their teachers, including inter-university-run learning opportunities and a virtual science laboratory.
• According to Check-a-Salary, the average salary for a materials scientist in the UK is around £35,000 per year. Prospects.ac.uk highlights that as a senior engineer with chartered status, you can earn up to £60,000 per year.
Meet Theo
My undergraduate degrees were in chemistry and chemical engineering. I then spent a summer interning in a cement manufacturing plant, before taking on a master’s degree in advanced process integration and design. It was my master’s project that really inspired me to follow the career path that I have.
Failure and mistakes are essential for development, especially for scientists. They’re an inevitability, but every setback presents an invaluable learning opportunity. Obstacles will come, but never give up.
The worst advice I have been given is to not be myself. Since then, colleagues, friends and family convinced me to follow my own ambitions, and this has been essential to my career.
My proudest career achievement was when I was awarded the UKRI Future Leaders Fellowship. On the theme of “Green, Circular, and Smart Cement Manufacture”, I’ll help lead research in low-carbon cement production. I also aim to use this research to influence UK policy and write standards for new innovative cements. Furthermore, I can use this platform to help mould other future leaders in my field.
Theo’s top tips
1. Surround yourself with people who motivate you.
2. Be true to yourself.
Do you have a question for Theo?
Write it in the comments box below and Theo will get back to you. (Remember, researchers are very busy people, so you may have to wait a few days.)













