How can scientists improve the well-being of patients with lupus?
Systemic lupus erythematosus (SLE) is a disease without a cure. But, thanks to discoveries by Professor George C. Tsokos, a rheumatologist at Harvard University in the US, new treatments are on their way to improve the quality of life for patients.
TALK LIKE A RHEUMATOLOGIST
Autoimmunity — when the immune system attacks healthy cells in the body
Epigenetic — environmental (rather than genetic) factors that affect how genes function
Immune system — your body’s defences against infection
Inflammation — a process by which the immune system fights invaders. Symptoms may include swelling and pain
Pathogen — something that causes disease, such as bacteria and viruses
Protein — a complex biological molecule that performs specific functions in the body
Rheumatology — the branch of medicine that studies and treats diseases that affect the muscles, bones, ligaments and tendons
T cell — a part of the immune system, trained to attack a specific pathogen
Tissue — a group of similar cells that work together as a unit, e.g., muscle tissue
Imagine your body is a castle. Your immune system is a team of guards, carefully patrolling the corridors to check for intruders. These guards are highly trained and know how to spot invading enemies, such as viruses or bacteria, and distinguish them from those who live in the castle, i.e., your own organs. When the guards encounter someone who should not be there, they quickly learn how to neutralise the threat, keeping the castle safe and secure.
But what if your guards turned against you? Imagine if, instead of attacking intruders, the guards started to fight the castle occupants. Not only would this cause damage and destruction to the castle, but it would leave the door open for enemies to sneak in.
Something similar can happen to a real human immune system, in a process known as autoimmunity. Instead of just attacking harmful viruses and bacteria, the immune system will sometimes turn on healthy cells in the body and attack them, too. This can result in autoimmune diseases such as Type 1 diabetes, rheumatoid arthritis and multiple sclerosis. Professor George C. Tsokos, a rheumatologist at Harvard Medical School and Beth Israel Deaconess Medical Center, has dedicated his career to researching one such autoimmune disease – systemic lupus erythematosus (SLE), a form of lupus.
What is lupus?
Although the name ‘lupus’ is Latin for ‘wolf’, the disease does not have much to do with wolves. It was named this in the 13th century, by a doctor who found some patients developed a rash on their face that looked like a wolf bite.
While cutaneous lupus only affects the skin, resulting in rashes and sores, SLE, the most common form of lupus, can affect any part of the body. The immune system in a person with SLE may attack any healthy tissue, including their internal organs, causing inflammation of the tissue and its eventual death.
People with lupus tend to have sore and swollen joints and feel very tired all the time, no matter how much they rest. It is also common for them to experience chest pain, rashes, headaches, fevers, swollen legs and hair loss, though different people will be affected by lupus in different ways.
As the disease can affect so many different body parts, and there are so many different symptoms, treatments must be tailored for each individual to help them manage their unique condition. While there is no cure for lupus, modern treatments have dramatically improved the well-being and life expectancy of people with the disease. Today, 80% of patients will live for more than 10 years after diagnosis. “Infections are the main cause of death for patients with SLE as the immune system struggles to recognise and fight pathogens,” explains George.
What causes lupus?
Although it is estimated that five million people worldwide live with lupus, the specific causes are still unknown. Unlike diseases such as COVID-19 and tuberculosis (TB), you cannot catch lupus from someone else, as it is not caused by viruses or bacteria. Instead, many scientists think lupus develops due to a combination of genetic and environmental factors.
“Nine out of ten lupus patients are female,” says George. “This signifies the importance of hormones and sex chromosomes in the expression of the disease.” Severe lupus is also more common in females of African, Hispanic and Asian descent, although it is unclear why. “We believe this is due to a combination of genetic and socioeconomic factors,” he explains. “The higher mortality in these demographics may be impacted by racial disparities in certain communities.”
What are T cells?
If your immune system is a team of guards, then your T cells are special agents. They are components of the immune system trained to fight pathogens, with each T cell specially equipped to recognise a particular invader of the body. T cells move around the body looking for the specific pathogen they have been trained to fight and, if they detect it, they attack any infected cells and communicate with the rest of the immune system to mount a response. However, when someone has an autoimmune disease, such as lupus, their T cells become confused. They lose their ability to discriminate between healthy cells and infected ones, so the immune system starts to fight healthy tissue, and the body essentially attacks itself.
How does George study lupus?
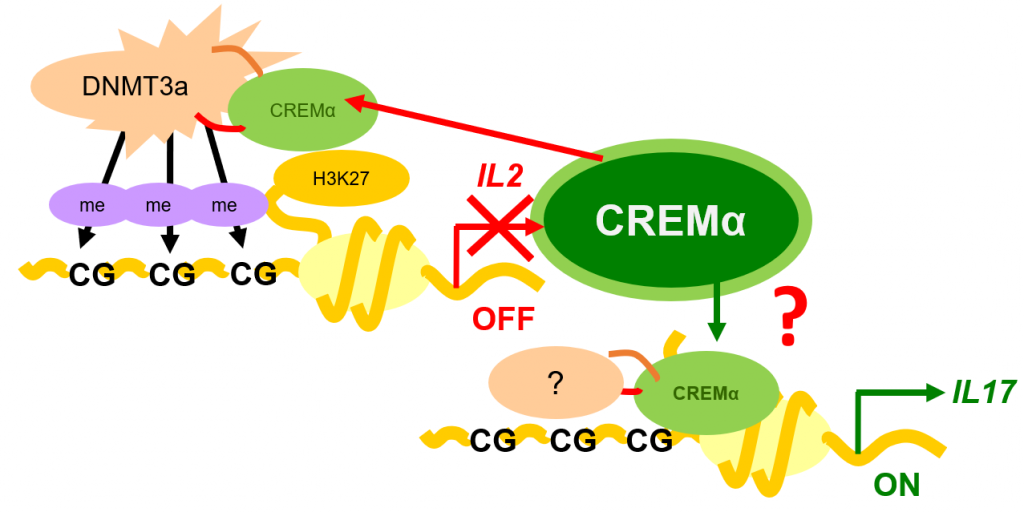
George and his research team take blood and tissue samples from patients with lupus to investigate which genes and molecules they contain. Through their research, they have discovered that the T cells in people with lupus do not produce a protein called interleukin 2 (IL-2), responsible for controlling inflammation, and produce too much of a protein called interleukin 17 (IL-17), responsible for causing inflammation.
Once the team establishes which molecules need further investigation, the researchers genetically engineer mice to produce the same molecules and test whether these mice develop lupus. This confirms whether the molecules of interest play a role in the development of the disease. After this confirmation, the team returns to patients to improve treatments based on their findings.
How is George’s research benefitting patients?
Reference
https://doi.org/10.33424/FUTURUM315
“One of the major discoveries in my lab has been the identification that a gene called CREM plays a role in protein expression in lupus patients,” says George. “We have shown that through distinct molecular and epigenetic processes, CREM suppresses the expression of IL-2 while it promotes the production of IL-17.” When mice were made to overexpress CREM, they did not produce IL-2 and produced more IL-17, while CREM-deficient mice did not develop autoimmunity. These findings are paving the way for new treatments for lupus patients.
George’s team has shown that administering low doses of IL-2 to mice with lupus reduces their level of autoimmunity and reduces their symptoms. Clinical trials are now in progress to test whether the same outcomes occur in humans. Early results appear promising, with low doses of IL-2 providing clinical benefits to patients with lupus.
The team has also shown that IL-17 contributes to organ inflammation in people and mice with lupus. “We have discovered that the T cells that produce IL-17 are present in the kidneys of people and mice with lupus,” explains George. As a result of this work, clinical trials are currently ongoing to test whether it is possible to block IL-17 in people with lupus, and whether this will deliver clinical benefits to them.
Other work has involved defining the molecular mechanisms that prevent lupus T cells fighting pathogens, leading to increased risk of infection in lupus patients and demonstrating that it is possible to restore the ability of these T cells to fight pathogens using precision nanotechnology approaches. By dedicating his career to studying lupus, George is improving the lives of the millions of people who live with this devastating disease.
 PROFESSOR GEORGE C. TSOKOS
PROFESSOR GEORGE C. TSOKOS
Chief of Rheumatology and Clinical Immunology, Beth Israel Deaconess Medical Center, Boston, USA
Professor of Medicine, Harvard Medical School, USA
Field of research: Rheumatology
Research project: Uncovering the molecular mechanisms that cause lupus in order to find new treatments for the disease
Funders: National Institutes of Health (NIH), Alliance for Lupus Research
ABOUT LUPUS RESEARCH
As one the world’s leading experts on lupus, George has uncovered key knowledge about the disease. However, there are still many questions about lupus that remain unanswered. “We need to focus on achieving a better understanding of the great heterogeneity of the disease as every patient experiences lupus in a different way,” he says. “We know that many mechanisms are involved, but we need to understand which ones are responsible for the development of the disease in each patient.” If this could be determined, it would enable improved precision medicine, whereby each patient could receive personalised treatment for their specific lupus conditions and symptoms.
Other aspects of lupus also need further exploration. We do not yet know what happens inside different organs to cause inflammation, or how the body’s central nervous system reacts to try and control this. Advances in technology are allowing scientists to study how remote elements in chromosomes interact to cause lupus and to investigate the molecular signatures of cells in various organs, which may shed light on some of these gaps in our understanding.
Could there be a cure for lupus one day?
The clues to how lupus manifests in patients could be hidden deep inside human DNA. Finding out exactly how certain genes lead to the disease will require many more years of careful study. Once we understand this, George thinks the knowledge could lead to a whole host of new treatments. “With the advent of gene editing tools, we may dream of offering such approaches to treat people with lupus,” he says. It is too soon to say whether a complete cure for lupus will be possible, but this approach could combat the genetic causes of the disease.
Who will help improve our understanding of lupus?
Improving the lives of people with lupus requires a team effort of scientists and clinicians in a range of fields. Molecular biologists and biochemists are needed to determine the reactions that occur within cells, geneticists are needed to uncover which genes are responsible for the disease, and scientists with skills in gene editing will attempt to prevent lupus from developing in people who may be susceptible.
Epidemiologists and social scientists are required to understand how socioeconomic factors influence the impacts of lupus in different populations. George believes that artificial intelligence will play an increasingly important role in studying lupus, so computer scientists and statisticians will also contribute to future research.
The healthcare professionals who work with patients with lupus are key to this mission. From general practice doctors to rheumatologists, nurses to clinical trial researchers, these people are the ones who will diagnose the disease, care for patients and test new treatments in the years to come. How could you help improve the lives of people with lupus?
Pathway from school to lupus research
• Study science subjects at school and college. A strong foundation in biology and chemistry is essential for understanding lupus, and knowledge of maths and statistics is necessary for analysing data.
• As improving our understanding and treatment of lupus requires scientists from a range of fields, many university degrees could lead to a career in the field. These include biochemistry, molecular biology, bioinformatics, genetics, biotechnology, epidemiology, public health and pharmacology.
• If you are interested in working directly with patients, you could study medicine or nursing to pursue a clinical career.
Explore careers in lupus research
• As with any disease, researching lupus requires working with people who have the disease, as they are the experts on how it impacts their life. The Lupus Foundation of America (LFA) has information about the range of research studies it is supporting, including current clinical trials for new lupus treatments: www.lupus.org/advancing-research/get-involved-in-research
• LFA also offers internships for university students: www.lupus.org/about-us/careers-and-internships
• Learn more about the research conducted in George’s lab: www.bidmc.org/research/research-by-department/medicine/rheumatology/tsokos-lab
Meet George
Who inspired you to become a doctor?
There was a doctor in my family, and, growing up, I had watched him work in his clinic and operating room. I liked the aura he displayed when helping people with their problems. I entered medical school in Athens, where I studied immunology, statistics and clinical medicine. I had the good luck to be taught by professors who were not only great clinicians, but also outstanding researchers. Listening to them talk about both clinical events and the underlying mechanisms made me want to become both a doctor and researcher.
And what led you to specialise in lupus?
My first patient in my first clinical rotation was a 24-year woman with lupus. The rest is history! I left Greece and continued my studies at the Arthritis Branch of the National Institutes of Health in the US. At that point, the Branch was dedicating most of its efforts to studying patients with lupus. I learnt a lot about lupus and started conducting research to understand the molecular development of the disease. My first project was to characterise the responses of T cells in patients with lupus. In the process, I had to learn more biochemistry, molecular biology, genetic engineering and bioinformatics.
What qualities do lupus researchers need?
I am driven by unsolved questions, and I strive to acquire the skills and knowledge to answer them. It is important to understand the great challenges faced by people with lupus (who are predominantly young women) and the need to regain their stolen lives. Lupus researchers can advance our knowledge of the disease through whichever field they love the most; the main requirement is a personal devotion to the care of people with chronic diseases.
What do your roles as a medical doctor and research scientist involve?
As the chief of rheumatology and clinical immunology at the Beth Israel Deaconess Medical Center, I oversee the clinical activities of the division. As a staff of 20 doctors and nurses, we see more than 25,000 patients each year. As well as treating patients in the hospital, I supervise the training of clinicians in rheumatology, help to lead the dedicated Lupus Center of Excellence, and teach students and trainees at BIDMC and Harvard Medical School.
I also run a laboratory with 15-30 researchers where we investigate the cellular, molecular, biochemical and metabolic aspects of lupus. We start by studying tissues from patients to identify which molecules are over- or under-expressed in people with lupus. We then genetically engineer lupus-prone mice to over- or under-express these same molecules, to demonstrate that these molecular abnormalities are important in living animals, before returning to the patients to conduct treatment studies.
What do you enjoy about your multiple roles?
Well, I find pleasure with everything I am doing. Seeing patients holds me to the ground. I get to see what people with lupus need and focus my efforts on solving their problems. Teaching is always exciting, particularly in a setting where students and young physicians inspire you with novel ideas. And research presents me with challenges to advance the field by asking new questions. I am excited by the continuous need to learn new approaches to solve problems.
What are your proudest career achievements?
I am proud to have advanced, even minimally, the field of lupus research and treatment by discovering novel mechanisms of the disease. It is also rewarding to know that the medical industry has picked up many of our findings and so they are now being applied in the clinic. And I am happy to have learnt from so many bright young colleagues who have moved on to build their own careers.
George’s top tips
1. Find a field that inspires you to dedicate significant effort.
2. Find questions that interest you, and stick with them until they are answered.
Do you have a question for George?
Write it in the comments box below and George will get back to you. (Remember, researchers are very busy people, so you may have to wait a few days.)












Hi my name is ashlee pillay and I am a south african Indian and very interested on getting some help for my partner who is currently 35 years old and has been living with lupus for 8 years now she is the most wonderful person iv ever came across and would love for her to enjoy her life,she has so much more to offer and I truly love her we want to actually start a family together and get married but because of this illness we don’t know where we ly, I would appreciate it if some one could look into helping us has I do know that one day she can do so much for others.please look into helping us because I don’t want to lose her and I know she as so much to offer the world,she is such a strong woman and goes through so much pain unfortunately the only thing u can do it massage her which dose not even help her at all it’s so hard to see her living in constant pain I do wish I could see her happy and enjoying her life as she is doing her best to remain happy and going the extra mile to fight just to stay Alive am happy iv meet her and she brings so much of joy into my life so please look into helping us on has u do know there is not much time left thanking you in advance ashlee